

ERT is one of several therapy options … please Click to call, text, or email us and we would be happy to help you to make sense of and prioritize your options.
Overview of Phase II ERT Clinical Trial
Additional study group for those under 18 months added to trial – May 2019
Discussion & Thoughts on this trial
Historical information – How we got here, Shire historical development updates, etc.
The following therapy and clinical trial descriptions were prepared by MLD Foundation. This page is continually updated and the content is copywritten.
Please do not copy this content, rather link to this page (https://MLDfoundation.org/ert/) when sharing this content so others always have the most up to date information.
Much like a diabetic receives regular insulin injections to manage their diabetes, Enzyme Replacement Therapy is an infusion (injection) of an enzyme designed to substitute for the missing ARSA enzyme that causes metachromatic leukodystrophy. ERT has been successfully used for other lysosomal diseases for well over a decade.
Over time the body filters out the injected enzyme so the infusion is repeated every week to keep the enzyme levels up and relatively consistent.
The actual infusion is via a small port (soft rubber-like button) that is placed under the skin, usually on the front/side of the patient.
ERT has a long history in the lysosomal disease space. Dr. Roscoe Brady proposed ERT in the 1960’s and was a pioneer in developing and advancing the therapy. The first commercial ERT was approved in 1991 for Gaucher disease. There are currently over a dozen approved 14 ERT therapies for various lysosomal disorders.
ERT takes effect almost immediately, however, the infusions are targeting the brain (central nervous system – CNS), not the peripheral nervous systems.
ERT requires weekly infusions … its goal is to stabilize the disease and prevent/minimize further progression, but it does require weekly visits to an infusion center, which for the clinical trial, is likely not in your home town. Eventually, there may be local and in-home infusion centers but that is not a formal part of the current trial offering.
ERT may not be an exclusion to participate in a future clinical trial or access a future approved therapy as the ERT can be stopped. Note well that this is a big may, but the hope is that ERT could be used to stop current progression and “buy time” for a more permanent or easier to maintain therapy.
The current trial includes a participation option for symptomatic patients, but participants showing symptoms may not show as much benefit aa those with little or no symptoms.
Takeda Pharmaceutical Company is developing the MLD ERT therapy, which they refer to as SHP-611. The initial development and clinical trial work were performed by Shire PLC, which was acquired by Takeda in early 2019. SHP-611 is based on several decades of Shire ERT research, expertise, and commercialization for other lysosomal disease products.
Registration – MLD ERT is still in Phase I/II clinical trial and has not been submitted for any agency review or registrations. As of Q3’2019, Takeda is forecasting regulatory approval and commercialization in 2023 based on the current clinical trial.
2012 Clinical Trial – A phase I/II dose-escalation study of 24 patients receiving HGT-1110 (Shire’s internal product reference code) with infusions every other week started in February 2012 and completed in January 2017. The trial analysis provided guidance to increase the dose frequency and amount for the current trial.
Orphan Designation – SHP-611 has been granted Orphan Designation by the EMA for Europe (EU/3/10/813.
Shire PLC had acquired an MLD ERT product called Metazyme from Zymenex in 2008. The clinical trial of Metazyme showed it was not effective.
A fifth study group – Group E – has been added to the inclusion matrix. Group E is for <18 month-olds with a GMFC-MLD score ranging from 1 to 4. The current Group D has a tighter inclusion criteria including being early symptomatic and a younger sibling of a trial participant.
You can learn more about the details of the trial including the now five study groups here. Or jump right to the detailed inclusion criteria here.
Takeda opened recruiting for their Phase II Clinical Trial for SHP611, an ERT therapy for MLD in April 2019. Recruiting was complete in January, 2021. Their marketing name for the study is the EMBOLDEN Study. You can learn more about the details of the trial including the four study groups here. Or jump right to the detailed inclusion criteria here.
The trial will include pre-symptomatic, and for the first time, symptomatic late infantiles (and their younger pre-symptomatic siblings). For participation with symptomatic children, please see the discussion below.
They have announced 24 sites, including for the first time, sites in the US. The sites will open one by one throughout 2019 and 2020.
RECRUITING was completed in January 2021. Below is a list of the active study sites.
United States
Please check back here for updated details and a list of open sites … Follow us on Facebook and subscribe to our Announcement Email list to keep up to date.
updated 5-Feb-2021
Takeda (formerly Shire) is in the midst of a Phase II Clinical Trial to expand its evaluation of the efficacy of their SHP611 Enzyme Replacement Therapy (ERT) for MLD. They call this study the “Embolden” study. The trial launched in April 2019 and completed enrollment of 42 patients in January 2021, 18 months before the target completion in the summer of 2022. The study lasts 210 weeks (4 years). Participants will complete the formal trial period starting mid 2023 and continuing through early 2024.
We expect some initial data reporting to occur before the formal end of the trial, but formal public reporting of full trial results will likely not be public until a year after the study completes, perhaps early in 2026. Patients will likely be offered continuing access to enzyme while the Embolden data is compiled, analyzed and reported.
Compared to the Phase I/II trial they have increased the dose, increased the frequency, will be opening sites around the world including for the first time in the US, and will be including symptomatic late-infantile patients.
The therapy is an Enzyme Replacement Therapy (ERT). Much like a diabetic receives regular insulin injections for diabetes, this is a weekly infusion (injection) of an enzyme designed to substitute for the body’s missing ARSA enzyme. ERT has been successfully used for other lysosomal diseases for over a decade.
 The drug delivery will be weekly through a small port (soft rubber-like button) that is placed under the skin via a minor surgery, usually on the front/side of the patient. From the port, a catheter (tube)
The drug delivery will be weekly through a small port (soft rubber-like button) that is placed under the skin via a minor surgery, usually on the front/side of the patient. From the port, a catheter (tube)
is connected to the spinal column which is a pathway for the enzyme infusions to flow freely into the brain. The cerebral spinal fluid (CSF) is on the other side of the blood-brain barrier and freely circulates through the spine to the brain. This permanent port eliminates the need for a lumbar puncture with each weekly infusion.
The infusions only take a few minutes, involve a quick needle prick into the port, and a bit of time post-injection to monitor for any reactions. The dose is 150mg (per kg), a higher and more frequent dose than the prior trial. The trial lasts 105 weeks (2 years) with an expectation that benefiting participants will be able to continue on the therapy after the formal trial ends as has been the case with the past studies.
The drug and all study-related testing will be paid for by the trial sponsor. Families should discuss the need for additional travel support, if any, at the time of assessment and enrollment.
Please feel free to contact us for up-to-date information on the trial, to do a pre-assessment of your loved-ones eligibility, to discuss expectations, and for assistance contacting the appropriate Takeda and investigative staff to be considered for the Clinical Trial. All formal trial inquiries will need to be made by your doctor directly to the Principal Investigator – we can help with that process. MLD Foundation has been working with Shire (now Takeda) since 2007 on this therapy and can quickly help you determine if your child might benefit from this study.
Recruiting for 35 patients in 4 four study groups started in April 2019 (now 5 groups with a total of 42 patients). Takeda launched with a small number of sites, including for the first time locations in the United States. They expect to expand to 24 sites worldwide 12-18 months. See above for a list of open study sites. You will be able to start the trial in one location and if a study location is closer to home opens then switch to that study site for the balance of the trial.
All study participants will be late infantiles, however, unlike previous phases, they will be recruiting children with varying degrees of disease progression, see table below.
The key inclusion criteria are:
click to jump to the detailed inclusion criteria
* Please contact us to discuss how your child may or may not fit these inclusion and exclusion criteria.
There are six groups of participants that are segmented by their GMFC-MLD scores.
• ERT does not repair existing myelin and nerve damage, rather, its goal is to dramatically slow or potentially stop further progression/damage.
• Pre- and very early symptomatic children will have the best long term outcomes. (Group A – GMFCS-MLD levels 1 & 2)
• It is not clear to us whether those showing advanced symptoms (groups B & C) when they start ERT will see much slowing of progression … see Discussion & Thoughts, below
• Group D is for younger affected siblings of participants enrolled in the trial.
Gross Motor Function Classification System or GMFCS is a 5 level clinical classification system that describes the gross motor function of people on the basis of self-initiated movement abilities. Particular emphasis on creating and maintaining the GMFCS scale levels rests on evaluating sitting, walking, and wheeled mobility. Distinctions between levels are based on functional abilities; the need for walkers, crutches, wheelchairs, or canes/walking sticks; and to a much lesser extent, the actual quality of movement.
The original version of the GMFCS was developed in 1997 cerebral palsy. The current GMFC-88 has been adapted to the unique characteristics of MLD and is referred to as GMFC-MLD.
The following three graphics may help you to preliminarily self-determine what GMFCS-MLD level your loved one might be. The 3rd graphic provides more detailed descriptions broken out by age group from birth to 18 years old.
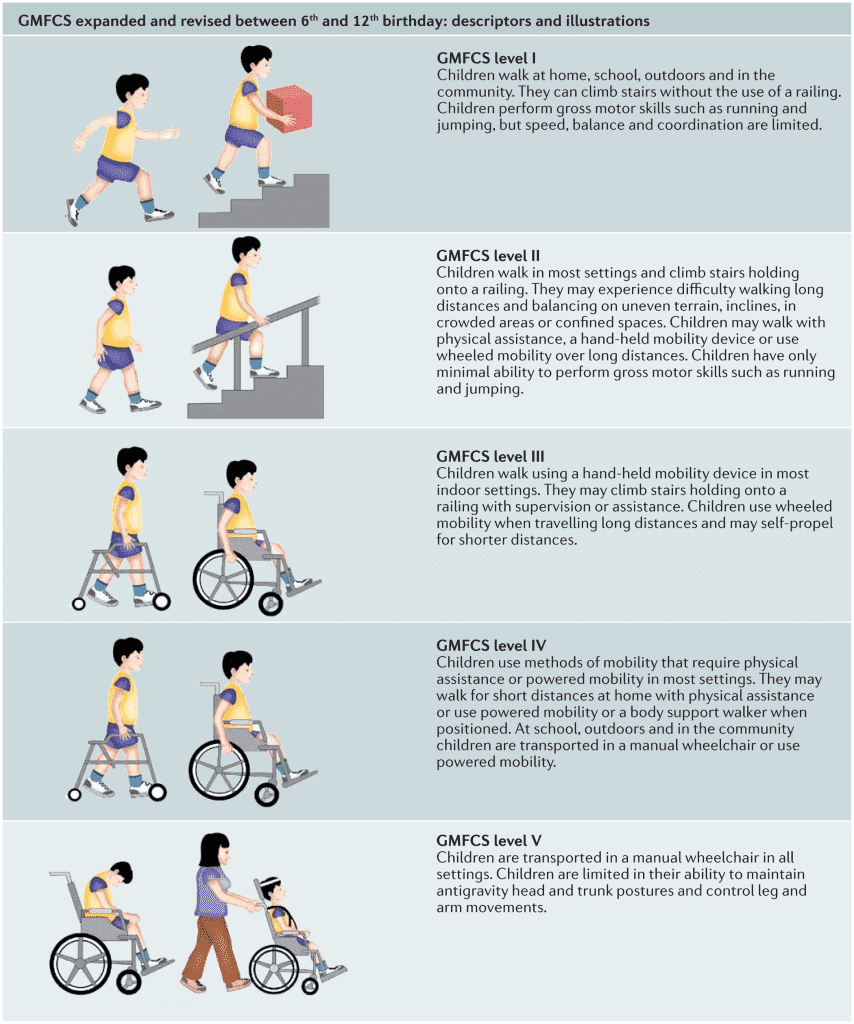
Click on the image below to enlarge it to show more details by age group from birth through age 18:
Study Summary – Weekly intrathecal injections for 105 weeks via an intrathecal drug delivery device (IDDD) or port implanted in the patient’s abdomen.
What is a Clinical trial? – Clinical trials are research studies performed in people that are aimed at evaluating a medical, surgical, or behavioral intervention. They are the primary way that researchers find out if a new treatment, like a new drug or medical device (for example, a pacemaker) is safe and effective in people. Efficacy, while desired and measured, cannot be guaranteed.
Infusion Time – The weekly infusion is expected to take just a few minutes, but it is likely patients will be monitored for an hour or two after each infusion to watch for any allergic reactions – which are not likely but is part of understanding the drug.
Cohorts – The study will be performed with 35 patients divided into four cohorts at varying degrees of disease progression.
Matched Historical Controls – these are untreated patients that will be identified through a separate CHOP natural history study and matched to treated patients based on medical history and progression. This enables Takeda to compare the efficacy of therapy to the natural history of untreated disease.
Enzyme – Recombinant human arylsulfatase A, which is expected to work in the same way as normal human arylsulfatase using a method known as “recombinant DNA technology”: it is made by human cells that have received a gene (DNA) that makes them able to produce arylsulfatase A.
 Intrathecal Port – The spinal fluid naturally circulates between the spinal column and the brain 3.7 times per day and it is on the “other side” of the blood-brain barrier that is a typical barrier to an intravenously delivered enzyme.* This intrathecal therapy delivers the enzyme directly into the spinal fluid. In order to minimize the chances of infection or injury due to repeated spinal puncture, a small port (a little larger than a US dime in diameter) will be implanted in the abdomen just under the skin – usually on the patient’s side or front which is connected via a very small catheter to spine and into the CSF.* The Enzyme is injected through the skin into the port and transfers via the catheter (tubing ) to the spinal fluid. This port is being studied as an integral part of the Clinical Trial. Please note – this is a static bolus device, not a pump. The image is the new (December 2013) Sophysa mini Soph-A-Port®.
Intrathecal Port – The spinal fluid naturally circulates between the spinal column and the brain 3.7 times per day and it is on the “other side” of the blood-brain barrier that is a typical barrier to an intravenously delivered enzyme.* This intrathecal therapy delivers the enzyme directly into the spinal fluid. In order to minimize the chances of infection or injury due to repeated spinal puncture, a small port (a little larger than a US dime in diameter) will be implanted in the abdomen just under the skin – usually on the patient’s side or front which is connected via a very small catheter to spine and into the CSF.* The Enzyme is injected through the skin into the port and transfers via the catheter (tubing ) to the spinal fluid. This port is being studied as an integral part of the Clinical Trial. Please note – this is a static bolus device, not a pump. The image is the new (December 2013) Sophysa mini Soph-A-Port®.
Placing the port and catheter is a minor surgery and will require sedation. Please ask us about how to best sedate MLD patients for this surgery.
24 International & US Sites – United States (California, Florida, Illinois, Iowa, Minnesota, New York, Ohio, Pennsylvania, Utah) Belgium, Brazil, Canada, Denmark, France, Germany, Israel, Japan, Mexico, Netherlands, Spain, United Kingdom.
You may enroll and start therapy at one site and then transfer to another site closer to home as it opens.
==> Details for each site, including contact information, can be found here.
Local Infusion Sites – Enrollment and periodic assessments must be at a primary study site (see above), however, it is Takeda’s desire to enable the weekly infusions to occur locally, if appropriate local resources are available. These local resources will have to be evaluated, authorized, and trained on a case by case basis.
Continuation of Enzyme Access After the Primary Study Period – Takeda has indicated they will continue to supply of enzyme after the 106 week study period. Historically this has been done as part of a separate “continuation study” that all patients are automatically enrolled in.
Primary End Points – Decline or lack of decline in GMFC-MLD. Group A is expected to decline no more than 2 levels. Additional observations and tests will be for Expressive Language Function Classification in Metachromatic Leukodystrophy (ELFC-MLD), change from baseline in Cerebrospinal Fluid (CSF) sulfatides levels, etc.
Compassionate Use/Named Access – to the best of our knowledge no Compassionate Use or Named Access for SHP611 will be made for non-trial participants during the recruitment phase of this study.
Phase II Trial Time Frame – Takeda estimates the recruiting and two year study period will end in late 2022.
Development Timeline – This ERT has been in various stages of Clinical Trial since 2012.
Why is the Trial Targeted for Late Infantile MLD? Most MLD Clinical Trials target the late-infantile form of the disease because this form of the disease is the most rapidly progressing and hence it is easier and faster to show the efficacy of the enzyme therapy.
Access for Juveniles and Adults – It is Takeda’s desire, after drug approval, to make the ERT available to patients with all forms and stages of MLD where efficacy can be expected. Studying the ERT in late-infantiles is the fasted path to therapy approval by regulatory agencies (FDA, EMA, etc.) Please note, your treating doctor (not Takeda) may determine that your loved one is too advanced to see any benefit from the therapy.
ClinicalTrials.gov – Trial ID NCT03771898
Takeda Embolden Study Overview – Trial Overview provided by Takeda
Orphan Designation – SHP611 has Orphan designation by the EMA for Europe (EU/3/10/813).
Pre-symptomatic and very early symptomatic late infantiles (Groups A, D, & E) – The primary outcome measures for this trial focus specifically on these groups, those with very little progression. Halting or dramatically slowing progression when there has been minimal damage will give the best long term outcomes. Group D is younger siblings of trial participants, most of which will likely be pre- or very early symptomatic.
Symptomatic Late Infantiles (Groups B, C, & F) – MLD Foundation believes the primary purpose of these groups is three-fold … to identify when a patient is too advanced for ERT to be effective, as a study arm to compare with Groups A, D, & E, and to provide some matched natural history control.
This is a clinical trial … and specifically with Groups B & C, it is a study to determine how and if ERT works when your child is progressed. Note that even with ERT there might still be disease progression in Groups B & C even if group A seems to hold more steady. As such, we think expectations for parents considering Group B & C enrollment should be well informed and appropriately tempered. We are not sure what, if any, expectations group F should have.
Over the past 20 years, we have met hundreds of families and their MLD loved ones in person, we’ve followed your journeys, and we have a good sense of how MLD progresses. We’ve also been working with institutional and biotech R&D, development, and clinical trials for over a decade. We have a lot of experience and perspective to share with you. These are tough decisions.
We know every family’s situation and considerations are different. Our hope is that you will be well informed before you make whatever decision you make. There is no right or wrong decision and we will support you regardless.
Feel free to call, email or text us to discuss your personal situation in more detail . We are happy to help.
© 2020, MLD Foundation, all rights reserved.